Day 2 Question 2: Millikan's Oil Droplet Experiment
Your solution: N:\oil\oil.{pas,c,cpp}
Input file: oil.in
Output file: oil.out
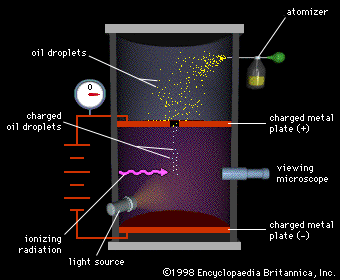 Millikan's famous Oil Droplet Experiment showed that electrical charge
is quantized; that is that the electrical charge on any object is the
sum of a number of elementary electrical charges. The electron is
the most common subatomic particle that carries a single elementary
electrical charge.
Millikan's famous Oil Droplet Experiment showed that electrical charge
is quantized; that is that the electrical charge on any object is the
sum of a number of elementary electrical charges. The electron is
the most common subatomic particle that carries a single elementary
electrical charge.
Millikan's experiment involved measuring the charge on several oil droplets,
and showing that each charge was a multiple of some smaller charge. Your
friend, the physics enthusiast, has to reproduce Millikan's experiment. She
has looked after constructing the apparatus and taking the measurements.
Her measurements are pretty good for what can be achieved in a
high school laboratory: the maximum error on any given
measurement is +/- 1%.
Given the measurements as input, your program must find the maximum possible value for
the elementary charge consistent with the measurements. That is, each
measurement, plus or minus an error of 1% or less, must be a multiple
of the elementary charge.
The input contains an integer n (1 <= n <= 100), the number of
oil droplets. This is followed by n real numbers on a single line; these represent the charge measured on each
droplet. The output should be a single number, the maximum possible
elementary charge, correct to 4 decimal places.
Sample Input
3
3.01 5.93 12.07
Output for Sample Input
2.9947
 Millikan's famous Oil Droplet Experiment showed that electrical charge
is quantized; that is that the electrical charge on any object is the
sum of a number of elementary electrical charges. The electron is
the most common subatomic particle that carries a single elementary
electrical charge.
Millikan's famous Oil Droplet Experiment showed that electrical charge
is quantized; that is that the electrical charge on any object is the
sum of a number of elementary electrical charges. The electron is
the most common subatomic particle that carries a single elementary
electrical charge.